
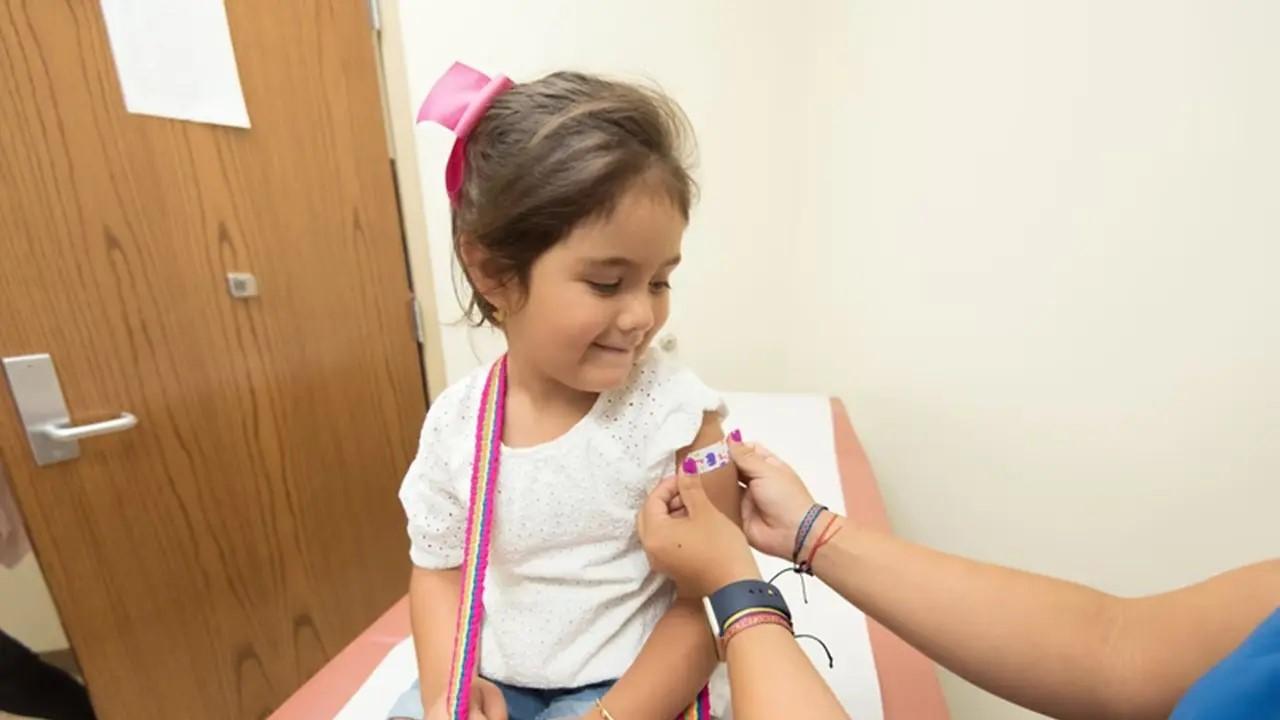
When Will Moderna's COVID-19 Vaccine Be Approved for Kids?
Moderna submitted a request in June for emergency use authorization of its two-shot COVID-19 vaccine for children aged 12–17. The FDA hasn’t given its approval yet.
The latest news in the healthcare sector including biotech and telehealth.






































© Copyright 2026 Market Realist. Market Realist is a registered trademark. All Rights Reserved. People may receive compensation for some links to products and services on this website. Offers may be subject to change without notice.