GlaxoSmithKline Has Developed a Broad Respiratory Portfolio
In 3Q17, GlaxoSmithKline (GSK) reported revenues close to 1.6 billion pounds from the sale of its respiratory products, which is year-over-year (or YoY) growth of 1% on a reported basis.
Jan. 26 2018, Updated 7:37 a.m. ET
GlaxoSmithKline’s respiratory franchise revenues
In 3Q17, GlaxoSmithKline (GSK) reported revenues close to 1.6 billion pounds from the sale of its respiratory products, which is year-over-year (or YoY) growth of 1% on a reported basis. The company witnessed sales worth 813 million pounds for its respiratory franchise in the US market in 3Q17, which is YoY growth of 1% on a reported basis and a drop of 1% on a constant exchange rate (or CER) basis. GlaxoSmithKline earned revenues close to 334 million pounds from the sale of respiratory products in Europe in 3Q17, which is a YoY rise of 2% on a reported basis and a drop of 1% on a CER basis. In 3Q17, the company witnessed respiratory segment sales close to 464 million pounds in international markets, which is YoY growth of 2% on a reported basis and 3% on a CER basis.
GlaxoSmithKline’s respiratory franchise earned revenues close to 5.1 billion pounds in the first nine months of 2017, which is 11% YoY growth on a reported basis and 3% growth on a CER basis. Revenues from the US market were close to 2.6 billion pounds, a YoY rise of 13% on a reported basis and 4% on a CER basis. The company also witnessed respiratory franchise sales worth 1.1 billion pounds in the European market, which is YoY growth of 5% on a reported basis but a 2% YoY drop on a CER basis. GlaxoSmithKline also earned revenues close to 1.5 billion pounds from the sale of its respiratory products in international markets in the first nine months of 2017, which is a YoY rise of 12% on a reported basis and 4% on a CER basis. GlaxoSmithKline makes up about 1.8% of the Vanguard FTSE Europe ETF’s (VGK) total portfolio holdings.
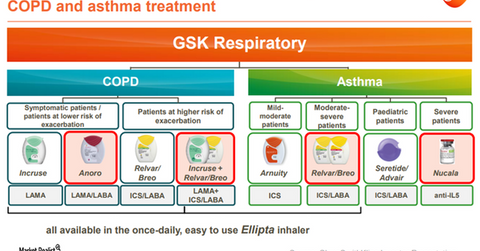
Broad respiratory portfolio
The above diagram gives a snapshot of the breadth of GlaxoSmithKline’s respiratory portfolio across various classes of drugs. With its recently launched products on the Ellipta platform and Nucala biologics, GlaxoSmithKline is competing in almost 80% of the respiratory drug classes. The Ellipta platform allows patients to take medications once a day, thus significantly improving chances of treatment adherence.
On February 23, 2017, GlaxoSmithKline announced results from its non-inferiority lung function study, which demonstrated that there was no impact on lung function if well-controlled asthma patients were switched from the company’s twice-a-day ICS/LABA inhaler, Advair/Seretide, to the recently launched once-a-day, Relvar/Breo Ellipta. These results may help GlaxoSmithKline compete effectively with other respiratory players such as Teva Pharmaceuticals (TEVA), Novartis (NVS), and Mylan (MYL) despite impending generic erosion for Advair/Seretide.
In the next article, we’ll discuss GlaxoSmithKline’s new product launches in the respiratory segment in greater detail.
