Atrial Fibrillation Ablation Could Be a Short-Term Growth Driver for MDT
More than 33 million patients suffer from AF, the most common form of heart arrhythmia. About 30% of these patients respond to antiarrhythmic drugs (or AAD).
Oct. 2 2017, Updated 7:41 a.m. ET
Atrial fibrillation ablation
Although its Implantable business is the largest part of Medtronic’s (MDT) Cardiac Rhythm and Heart Failure Disease Management (or CRHF) segment, diagnostics, infection control, atrial fibrillation (or AF) ablation, and other services are expected to be major short-term growth drivers for the company.
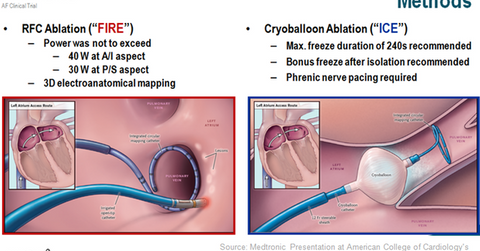
FIRE & ICE trial
In the FIRE & ICE clinical trial, Medtronic demonstrated the non-inferiority of cryoballoon ablation to radiofrequency current (or RFC) ablation in symptomatic patients suffering from paroxysmal AF. The procedure time also was significantly lower for cryoballoon ablation.
More than 33 million patients suffer from AF, the most common form of heart arrhythmia, and ~30% of these patients respond to antiarrhythmic drugs (or AAD). Around 40% of the AF patients are asymptomatic.
For the remaining 30% AF patients who are either refractory or are unable to tolerate more than one Class I or Class III AAD and who are also symptomatic, catheter ablation is the Class I Level A recommendation. Medtronic projects that only 4% of the ablation candidate patients are treated every year. This highlights the extent of market opportunity for Medtronic in the AF Ablation segment.
To leverage this opportunity, Medtronic has been working to develop a shorter tip for its AF cryoablation balloon systems. Coupled with Medtronic’s advanced mapping catheter, this is expected to improved device capability to measure transthoracic electrical impedance (or TTI), a major parameter for defibrillation efficiency.
With these innovations, Medtronic is expected to pose solid competition to cardiovascular device players such as Abbott Laboratories (ABT), Boston Scientific (BSX), and St. Jude Medical (STJ).
Medtronic accounts for 0.79% of the Vanguard Total Stock Market ETF’s (VTI) total portfolio holdings.
In the next article, we’ll discuss the growth prospects for Medtronic in Diagnostics space.
