Bristol-Myers Squibb’s Profitability: Can Eliquis Do the Job in 2017?
BMY’s Eliquis can be considered a leading therapy for US patients suffering from Afib (atrial fibrillation) and venous thromboembolism.
Jan. 25 2017, Updated 3:05 p.m. ET
Eliquis annual performance
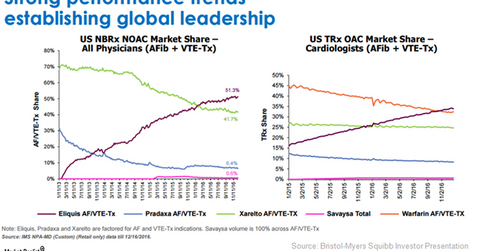
Based on the share of TRx (total prescriptions) for OAC (oral anticoagulants) written by cardiologists in 2016, Eliquis can be considered a leading therapy for US patients suffering from Afib (atrial fibrillation) and venous thromboembolism. Developed by Pfizer (PFE) and Bristol-Myers Squibb (BMY), the drug has surpassed other OACs like Pradaxa, Johnson & Johnson’s (JNJ) Xarelto, Savaysa, and Warfarin in terms of TRx market share.
For more on Eliquis’s growth drivers, please refer to Market Realist’s “Eliquis Proving to Be a Major Growth Driver for Pfizer This Year.” Notably, the iShares Core S&P 500 ETF (IVV) has about 0.43% of its total portfolio in BMY.
Growth drivers
In the US, Eliquis has managed to become the most preferred oral anticoagulant in the institutional setting. The drug has also been increasingly prescribed by primary care physicians. Eliquis’ share in the NBRx (new to brand) NOAC (novel oral anticoagulant) is currently around 51.3%, which is significantly higher than that of Xarelto’s 41.7% market share.
Bristol-Myers Squibb’s plans to target patients treated with warfarin for the future market expansion of Eliquis.
Eliquis has also been witnessing strong demand in major markets like Germany and Japan. Eliquis has thus managed to outperform branded competitors as well as generic competitors from companies like Teva Pharmaceutical (TEVA) and Mylan. For more on this, check out “Bristol-Myers Capitalizes on Japanese Markets for Its Cardio Drug.”
In the next part, we’ll discuss Bristol-Myers Squibb’s immuno-oncology strategy for 2017.
