JAVELIN Series: Clinical Studies of Avelumab
The JAVELIN series is a clinical trial program for the use of Avelumab for treatment of multiple types of cancer, including Merkel cell carcinoma.
Nov. 23 2015, Published 2:34 p.m. ET
Avelumab and the Merck-Pfizer alliance
As discussed earlier, Avelumab is an anti-PD-L1 blocking antibody being jointly developed as part of an alliance between Merck KGaA, Germany and Pfizer (PFE).
Both of these companies will jointly develop and commercialize anti PD-L1 antibody for the treatment of multiple types of cancer. This alliance will also strengthen the position of both these companies in the immuno-oncology segment.
JAVELIN Merkel 200: Study of Avelumab for Merkel cell carcinoma
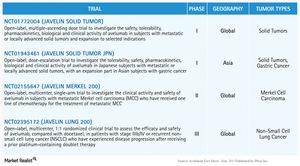
As per the information from Pfizer’s fact sheet dated May 19, 2015, the JAVELIN series is a clinical trial program for the use of Avelumab for treatment of multiple types of cancer. For Merkel cell carcinoma, the study is referred to as JAVELIN Merkel 200.
This study is presently in Phase II and is investigating the use of Avelumab for treatment of Merkel cell carcinoma for patients who have undergone at least one chemotherapy treatment.
The results of this study are not available. However, findings of this study have already helped the Merck-Pfizer alliance to fetch fast track and orphan drug designations for Avelumab over the last couple of months. The designation of breakthrough therapy for Avelumab is an achievement for both of these companies.
Other JAVELIN studies
Apart from Merkel cell carcinoma, the JAVELIN series is investigating the use of Avelumab in solid tumors, gastric cancer, and non-small cell lung cancer (or NSCLC).
For solid tumors, the study is termed JAVELIN Solid Tumor globally, and Javelin Solid Tumor JPN for Japanese patients. These studies are in Phase I at present. JAVELIN Solid Tumor JPN also has an expansion, which is focused on investigating the use of Avelumab in Asian patients with gastric cancer. For lung cancer, the study termed JAVELIN Lung 200 is presently in Phase III.
Other drugs used for the treatment of skin cancers such as melanoma include Merck & Co.’s (MRK) Keytruda, Novartis’s (NVS) Mekinist, Roche’s Cotellic, and Bristol-Myers Squibb’s (BMY) Opdivo.
In order to divest risk, investors can consider ETFs such as the iShares US Pharmaceuticals ETF (IHE), which holds ~9.5% of its total investments in Pfizer and ~8.4% of its total investments in Merck & Co.
