The Company Behind the Monkeypox Vaccine as Cases Rise Worldwide
There’s already a monkeypox vaccine, but health officials say the general public may not need it. Here’s what to know about the company behind the shot.
May 24 2022, Published 1:17 p.m. ET
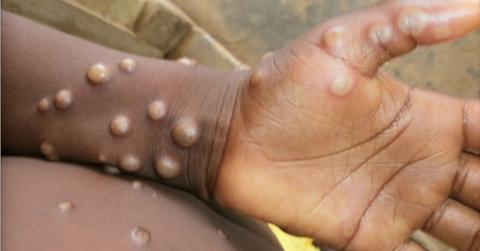
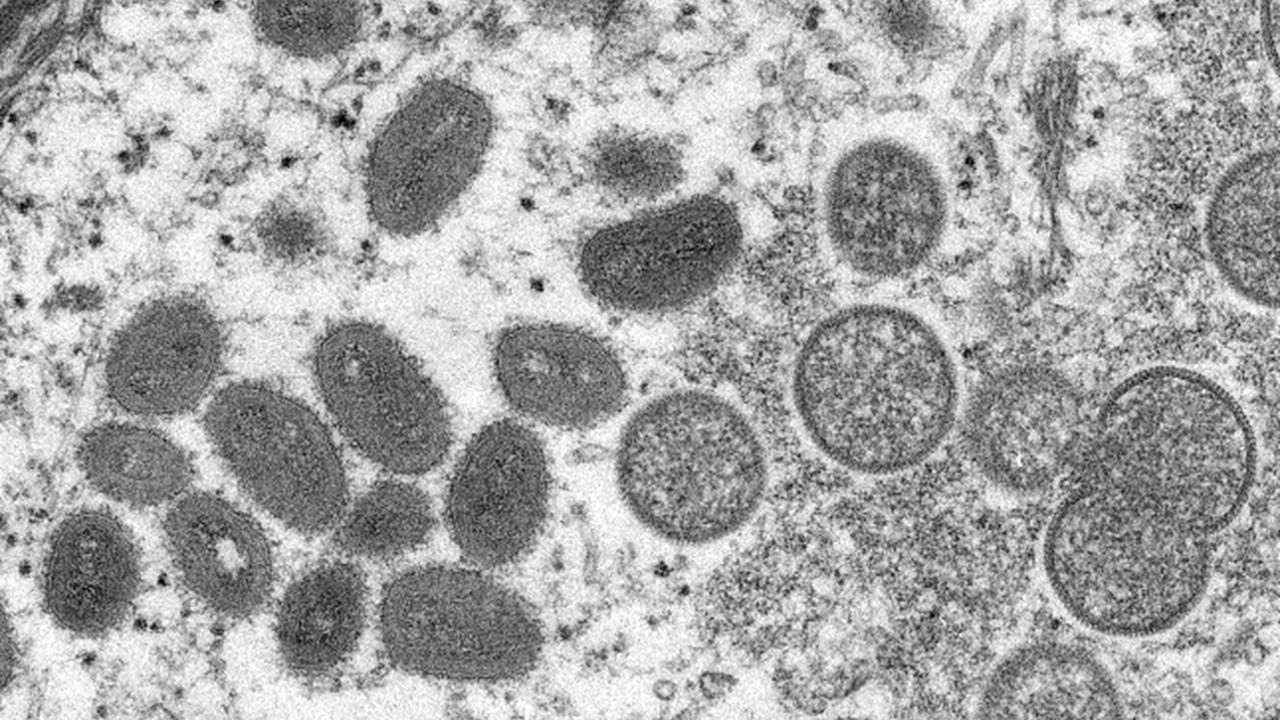
The World Health Organization (WHO) shared last week that the monkeypox virus is spreading globally. So far, 12 nations where monkeypox isn't endemic have reported cases, with the U.S. seeing upwards of five cases in the last year. There’s already a company producing a monkeypox vaccine, and the U.S. has its own supply.
Here’s what to know about the company behind the Jyennos monkeypox vaccine, which became FDA-approved back in 2019.
The FDA approved the Jynneos monkeypox vaccine in 2019.
The FDA approved a monkeypox vaccine called Jynneos in 2019. The vaccine even works after exposure to the virus, which makes it more effective at treating this rare but serious condition.
Monkeypox is endemic (regularly found among people) in Central and West Africa. Now, global cases are beginning to pop up even from people who haven't traveled to endemic regions. This is inflating the concern for monkeypox.
Who makes Jynneos?

Bavarian Nordic CEO Paul Chaplin
Jynneos (also called Imvamune or Imvanex) is a two-dose monkeypox vaccine suited for people 18 years of age or older. The same vaccine also works to prevent smallpox. The company behind the monkeypox vaccine is called Bavarian Nordic A/S.
Bavarian Nordic is a public company trading on the over-the-counter market under the ticker symbol “BVNRY.” The company is headquartered in Denmark and spearheaded by president and CEO Paul Chaplin. Chaplin has been at the helm since 1999.
With the concerning rise of monkeypox cases throughout the world, BVNRY stock has soared. Shares are up 34.08 percent in the five business days ending May 24 — and that’s after correcting a nearly 80-percent leap.
Bavarian Nordic’s other vaccine solutions include Rabipur (also called RabAvert, a rabies vaccine for human use), Encepur (a tick-borne encephalitis vaccine), and Mvabea (an ebola vaccine licensed to Janssen).
The U.S. will tap into the Jynneos monkeypox vaccine stockpile.
The U.S. has about 1,000 doses of the monkeypox vaccine currently in its stockpile. These will be reserved for high-risk contacts of early patients, according to Dr. Jennifer McQuiston, deputy director of the Centers for Disease Control and Prevention’s Division of High Consequence Pathogens and Pathology.
McQuiston added, “We expect that level to ramp up very quickly in the coming weeks as the company provides more doses to us.”
Do any other companies that make a monkeypox vaccine?
The U.S. also reportedly has 100 million doses of a smallpox vaccine called ACAM2000. Sanofi Pastuer Biologics Co. creates this vaccine, which is considered of an older generation and carries additional side effects. McQuiston discussed ACAM2000 and said, “A decision to use that widely would have to have some serious discussion behind it.”
Until then, Bavarian Nordic’s Jynnea monkeypox vaccine remains the primary solution for the ongoing monkeypox outbreak.
Investors are also rallying for stocks like GeoVax Labs Inc. (GOVX), Emergent Biosolutions Inc. (EBX), Inovio Pharmaceuticals Inc. (INO), and more. These were favorites in the early days of the COVID-19 pandemic when investors were pushing capital into vaccine researchers. None of them have a monkeypox vaccine, but that could change in the future.