Biogen’s Hemophilia Portfolio: Growing Market Share in 2Q15
Biogen’s (BIIB) hemophilia drugs, Alprolix and Eloctate, continued to gain new patients in 2Q15.
July 30 2015, Updated 11:07 a.m. ET
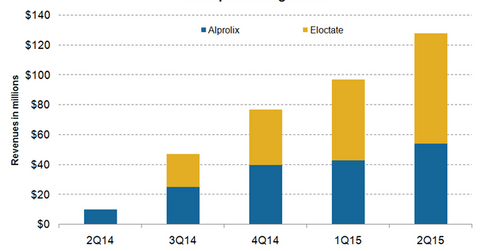
Hemophilia portfolio
Biogen’s (BIIB) hemophilia drugs, Alprolix and Eloctate, continued to gain new patients in 2Q15. According to the National Hemophilia Foundation, about 400,000 people in the world are afflicted by hemophilia.
In 2014, Biogen introduced two new drugs to the United States: Alprolix for hemophilia B and Eloctate for hemophilia A. Both drugs are considered to be a substantial advancement over pre-existing hemophilia drugs such as Pfizer’s (PFE) Xyntha and Baxter’s (BAX) Advate. New hemophilia drugs, however, are being developed by competitors such as BioMarin Pharmaceuticals’ (BMRN) BMRN-270 and Baxter’s BAX-335, increasing competition in the market. To learn more about Biogen’s hemophilia portfolio, please refer to Biogen’s Hemophilia and Alzheimer’s Disease Portfolio.
Alprolix sales
Compared to 2Q14, Alprolix registered revenue growth of 440% from $10 million in 2Q14 to $54 million in 2Q15. Revenues from sales in the US market rose by about 390% from $10 million to $49 million while revenues from sales in the rest of the world (or ROW) reached $5 million. ROW sales were primarily from Japan.
Biogen has filed to obtain marketing authorization for Alprolix in Europe. Biogen had added Alprolix to its product portfolio through the acquisition of BIH, formerly known as Syntonix, in January 2007. If this drug obtains EMA before January 2017, Biogen would have to make a contingent payment of $20 million to BIH.
Biogen entered the hemophilia market by collaborating with Swedish Orphan Biovitrum (or Sobi) to jointly develop Alprolix and Eloctate. On July 16, 2015, Sobi exercised its option to assume final development and commercialization rights for Alprolix in Europe, Russia, Turkey, and some countries in the Middle East. Biogen holds these rights in North America and the rest of the world, except Sobi’s territories.
Eloctate sales
Eloctate was introduced to the US market in 3Q14. Eloctate registered revenue growth of 37% from $54 million in 1Q15 to $74 million in 2Q15. Both Alprolix and Eloctate are currently undergoing testing for efficacy and safety in children below 12 years of age.
Investors can get exposure to Biogen’s upcoming hemophilia portfolio while reducing company-specific risks by investing in the iShares NASDAQ Biotechnology ETF (IBB). IBB holds 7.48% in Biogen stock.
