DexCom Inc
Latest DexCom Inc News and Updates

Tandem Diabetes Care’s International Expansion Strategy
Tandem Diabetes Care (TNDM) is slated to begin its international expansion later this year to capture opportunities arising after Johnson & Johnson’s (JNJ) exit from the insulin pump market, which was announced in October 2017.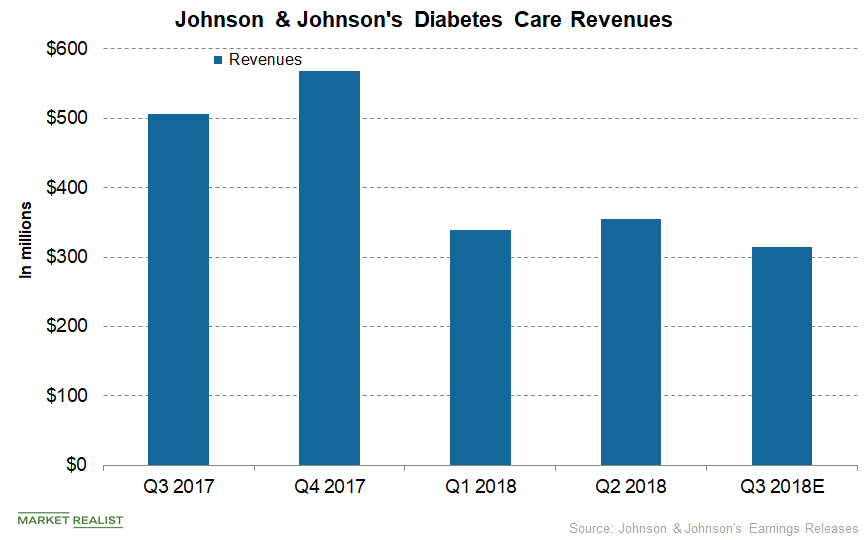
A Look into Johnson & Johnson’s Diabetes Care Business
In the US and international markets, Johnson & Johnson’s Diabetes Care segment generated third-quarter net revenues of $125.0 million and $190.0 million, respectively.
What to Expect from Tandem’s Product Pipeline
T:sport is Tandem’s next-generation hardware platform, which is expected to reduce the size of the t:slim pump by 50%.
Medtronic Strengthens Diabetes Business with MiniMed Infusion Set
On February 21, 2018, Medtronic (MDT) announced the launch of its MiniMed Mio Advance infusion set.
Abbott’s Breakthrough CGM Device Freestyle Libre Wins FDA Approval
On September 27, Abbott Laboratories (ABT) announced the FDA approval of its Flash CGM (continuous glucose monitoring) device, Freestyle Libre.Company & Industry Overviews What Explains the Columbia Select Large Cap Growth Fund’s Poor Showing?
The short-term performance of the Columbia Select Large Cap Growth Fund – Class A (ELGAX) is excellent, and the past six months have been great for the fund.
