Tasigna Could Boost Novartis’s Revenue Growth in 2018
In 1Q17, 2Q17, and 3Q17, Tasigna generated revenues of $411 million, $463 million, and $482 million, respectively.
Jan. 25 2018, Updated 9:00 a.m. ET
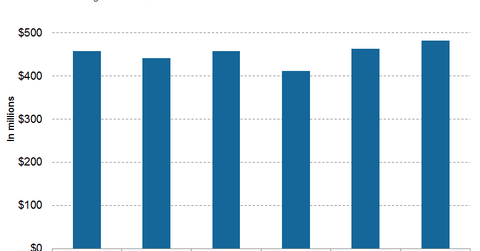
Tasigna revenue trends
In 1Q17, 2Q17, and 3Q17, Tasigna generated revenues of $411 million, $463 million, and $482 million, respectively. In 3Q17, Tasigna witnessed 9% growth on a year-over-year basis and 4% growth quarter-over-quarter.
Recent developments
In December 2017, the U.S. FDA (Food and Drug Administration) approved the inclusion of treatment-free remission (or TFR) data in Novartis’s (NVS) Tasigna product label.
Currently, Tasigna is the first and only BCR-ABL tyrosine kinase inhibitor (or TKI) to include data on attempting treatment discontinuation in qualified adults with Philadelphia chromosome-positive chronic myeloid leukemia (or CML) in the chronic phase after reaching a deep molecular response of MR4.5 in its FDA-approved prescribing information.
In the clinical trials, nearly half of the Philadelphia chromosome-positive CML patients in the chronic phase who discontinued Tasigna continued in TFR approximately two years after stopping therapy.
ENESTfreedom trial
Novartis conducted the phase 2 ENESTfreedom trial to evaluate stopping therapy in 190 adults with Philadelphia chromosome-positive patients in the chronic phase who received Tasigna as first-line therapy and sustained a deep molecular response for one year before discontinuation of therapy.
The ENESTfreedom trial found that 48.9% of patients out of 190 CML patients could discontinue therapy and continue MMR at 96 weeks. Among the 88 patients who restarted Tasigna therapy due to loss of MMR by the cut-off date, 87 patients regained MMR.
ENESTop trial
Novartis conducted the ENESTop trial, which evaluated stopping of Tasigna therapy in 163 Philadelphia chromosome-positive patients in chronic phase who were previously treated with Gleevec and then switched to Tasigna and achieved sustained molecular response for one year before Tasigna therapy.
In the ENESTop trial, 53.2% of patients were able to maintain TFR at 96 weeks. In the trial, 56 patients who confirmed a loss of MMR restarted Tasigna by the cut-off date. Among these patients, 92.9% of patients regained both MR4.0 and MR4.5.
Tasigna’s competitors include Bristol-Myers Squibb’s (BMY) Sprycel, Takeda Pharmaceuticals’ (TKPYY) Iclusig, and Pfizer’s (PFE) Bosulif. The revenue growth of Novartis’s Tasigna could boost the Pharmaceutical ETF (PPH). Novartis makes up about 4.7% of PPH’s total portfolio holding.
