MitraClip Drives Growth, Gets Reimbursement Approval in Japan
On March 19, 2018, Abbott Laboratories (ABT) announced that it had received reimbursement approval for its MitraClip device in Japan.
April 9 2018, Updated 12:35 p.m. ET
MitraClip receives reimbursement approval in Japan
On March 19, 2018, Abbott Laboratories (ABT) announced that it had received reimbursement approval for its MitraClip device in Japan.
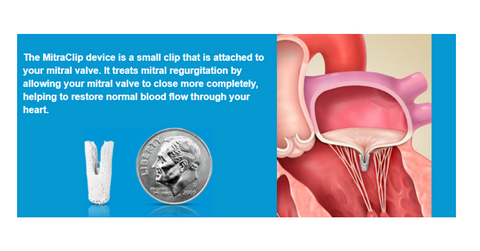
MitraClip is a minimally invasive procedure for patients suffering from MR (mitral regurgitation) who are at high risk of open surgery. Japan’s MHLW (Ministry of Health, Labour and Welfare) granted Abbott’s MitraClip national reimbursement approval for the treatment of MR patients.
Abbott’s MitraClip approval
In November 2017, MitraClip was approved as the first TMVR (transcatheter mitral valve repair) device in Japan for the treatment of severe DMR (degenerative mitral regurgitation) and FMR (functional mitral regurgitation) heart diseases. The abnormality of valve structures, including valve ring, leaflets, and chordae tendineae, result in DMR, while FMR is caused by the mitral valve’s incomplete coaptation due to the dilation of the left ventricle.
The device was approved in the United States in 2013 and received CE mark approval in Europe in 2008. To date, Abbott’s MitraClip has been used to treat more than 50,000 people across nearly 50 countries. Medtronic (MDT), Edwards Lifesciences (EW), and Boston Scientific (BSX) are Abbott’s major competitors in this market.
The reimbursement approval in Japan became effective on April 1, 2018. It renders the minimally invasive MR therapy using MitraClip more accessible to patients in the country.
Management’s view
According to Michael Dale, vice president for Abbott’s structural heart business, “Our mission is to develop technologies and therapies that help people return to better health and quality-of-life as quickly as possible. With this reimbursement in Japan, we can help more people live better by reducing the severity of an extremely life-altering illness in a safe and predictable way.”
In the next article, we’ll look at analysts’ latest recommendations and target prices for Abbott.
