Fully Funded Partnerships May Drive Growth for Ligand Pharmaceuticals
Ligand Pharmaceuticals (LGND) expects its licensees to invest ~$2.0 billion for advancing more than 155 partnered research and development programs in 2017.
Aug. 31 2017, Updated 9:07 a.m. ET
Fully funded partnerships
Ligand Pharmaceuticals (LGND) expects its licensees to invest ~$2.0 billion for advancing more than 155 partnered research and development programs in 2017. These involve investments in 32 Phase 3 trials, 39 Phase 2 trials, 46 Phase 1 trials, four Phase 4 trials, and 91 pre-clinical trials, as well as in scaling up manufacturing facilities and fees for regulatory filings.
Ligand Pharmaceuticals has ~92 partners and expects ~$2.0 billion in milestone payments from these programs. Ligand Pharmaceuticals expects the number of commercialized products generating revenues to rise from 14 in 2016 to more than 28 in 2020. Ligand Pharmaceuticals makes up about 0.69% of the iShares S&P Small-Cap 600 Growth ETF (IJT).
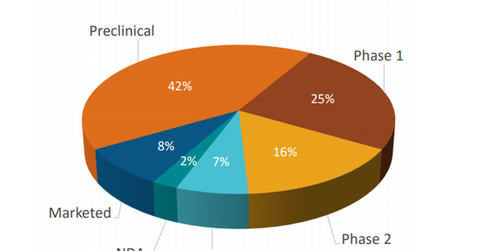
Research program distribution
The chart above shows the percentage distribution of Ligand Pharmaceuticals’ research programs across various stages of clinical development. Around 10% of the research programs are either in the already marketed or new drug application (or NDA) stage.
Ligand Pharmaceuticals is well positioned to compete effectively with peers such as Jazz Pharmaceuticals (JAZZ), Ionis Pharmaceuticals (IONS), and United Therapeutics (UTHR) for investor attention.
Ligand Pharmaceuticals’ research programs span multiple therapeutic indications, with exposure to each indication roughly equal to the total spending for that area in the pharmaceutical industry. The company has 33% of its programs focused on oncology indications, and 25% of its programs target diseases of the central nervous system, autoimmune diseases, and infectious diseases.
Other research programs are focused on indications in areas such as antiviral, cardiovascular, dermatology, genetic disease, hematology, and inflammatory diseases. These programs also address areas covering medical devices, metabolic diseases, musculoskeletal, ophthalmology, respiratory, and women’s health conditions.
In the next article, we will discuss Ligand Pharmaceuticals’ research and development strategy in greater detail.
